Prime Global’s Sue O’Leary and Jenny Blackham were pleased to host this roundtable at the recent World Pharma Pricing Market Access & Evidence Congress. Here’s the highlights from their session:
read more >>Part Two: Insights from the 16th Annual Meeting of ISMPP 2020: The Evolving Role of The Scientific Communications Professional in an Open World
Prime Global 27 August, 2020We were proud to exhibit at ISMPP’s first virtual Annual Meeting and contribute to useful discussions about the evolving role of the scientific communications professionals. Our team enjoyed interacting with attendees and gathering insights from a brilliant agenda. To read more about the sessions, view our insights here:
read more >>Part One: Insights from the 16th Annual Meeting of ISMPP 2020: The Evolving Role of The Scientific Communications Professional in an Open World
Prime Global 12 August, 2020We were proud to exhibit at ISMPP’s first virtual Annual Meeting and contribute to useful discussions about the evolving role of the scientific communications professionals. Our team enjoyed interacting with attendees and gathering insights from a brilliant agenda. To read more about the sessions, view our insights here.
read more >>European ISMPP 2018
Neil Venn 25 January, 2018Thank you so much to ISMPP (International Society for Medical Publication Professionals) for organising another brilliant EU event for 2018. Our highlight was our own Prime Person, Neil Venn, presenting our research on, “Do authors fully utilize opportunities to share supplementary information at conferences?” #ismppeu18 #ismpp #science #publications #research
read more >>End to a successful ISMPP meeting
Emily Anslow 5 May, 2017Following successful #ISMPP meeting welcome drinks for Akua and Shaunda in the New York Prime Global #MedComms office
read more >>We really enjoyed seeing old friends and making new ones. Our highlights were the dynamic posters and the discussions around them. Thanks to everyone who came to visit us at our booth, and of course to ISMPP for organising. See you next year!
read more >>Day 2 of #ISMPP is under way!
Aisleen Peterson 2 May, 2017We’re especially looking forward to the Publishers Panel: Simplifying and Streamlining – Working Together to Improve the Quality of Medical Publications for some critical insights and new perspectives. ISMPP (International Society for Medical Publication Professionals
read more >>ISMPP Day 1 Highlights
Aisleen Peterson 2 May, 2017Meeting new people, catching up with old faces and discussing the brilliant array of posters submitted this year. – Our very own Neil Venn presenting Prime Global’s poster on ‘Sharing Enhanced Poster Content’ – a fascinating discussion chaired by Neil! ISMPP Day 1 Key Learning: Deborah A. Zarin (Director of ClinicalTrials.gov) gave an excellent talk on “The Role of ClinicalTrials.gov in an Era of Data Sharing”, providing an overview of the history of clinicaltrials.gov, as well as information on the impact of recent guidelines which require the uploading of clinical trial information and data in a timely fashion.
read more >>Patricia Baskin from #CScienceEditors at #ISMPP talk about predatory journals. Important to follow the check list https://lnkd.in/gF4vqS8
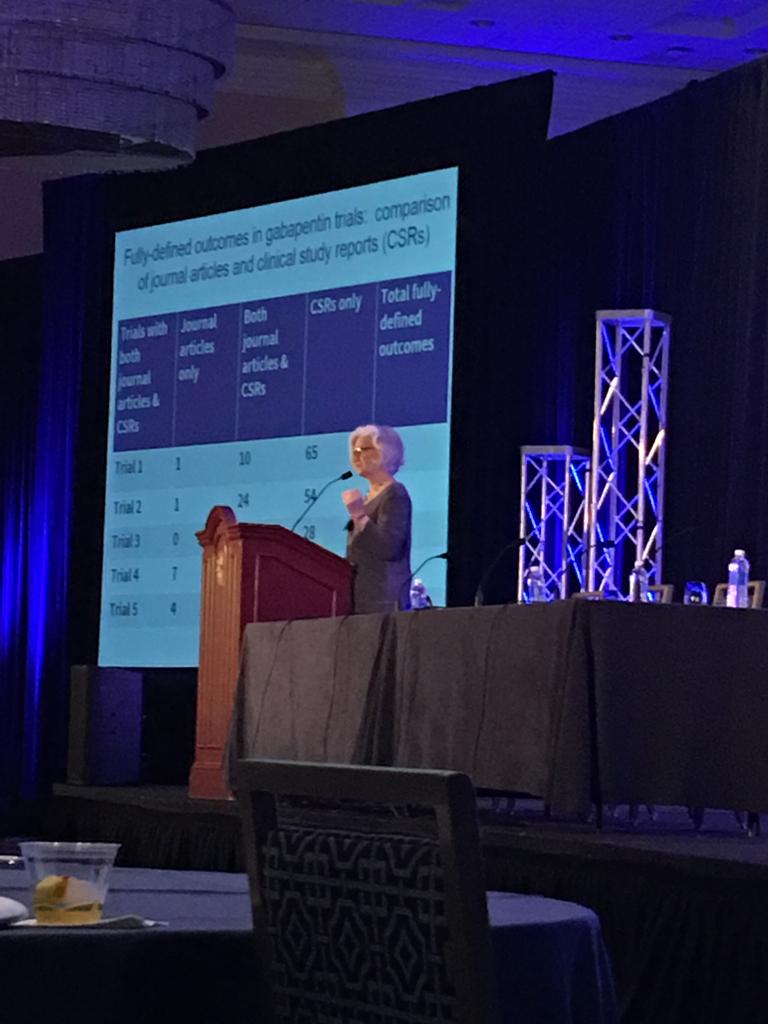
read more >>@KayDickersin keynote about clinical trial reproducibility and outcomes.
read more >>Enhanced poster content
Aisleen Peterson 2 May, 2017Great discussion around sharing enhanced poster content.
read more >>ISMPP
Aisleen Peterson 1 May, 2017Valerie leading a roundtable session.
read more >>IMSPP
Graeme Peterson 1 May, 2017We’re excited for the next 3 days at #ISMPP. Remember to visit our booth #24 to speak with our experts #medcomms
read more >>Looking forward to ISMPP
Graeme Peterson 1 May, 2017Looking forward to a lively discussion at ISMPP
#ISMPP #MedComms #Publications #ScientificPubs
read more >>Our San Francisco Team
Aisleen Peterson 29 April, 2017A brilliant shot of some of the San Francisco team enjoying the views at the Top of the Mark ahead of ISMPP — Only 2 days to go!
read more >>Gearing up for ISMPP
Aisleen Peterson 28 April, 2017Brilliant to see some of our New York and Knutsford colleagues catching up over dinner in the Big Apple as they prepare for ISMPP. Cheers guys, wish we were there!
read more >>ISMPP meeting in Washington
Aisleen Peterson 18 April, 2017We’re really looking forward to the annual ISMPP meeting in Washington – it’s only two weeks away. We’ll be covering a round-table session and finding out all the latest news and trends in #publications. Hope to see you there.
read more >>